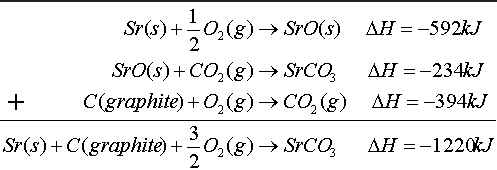Chapter 10--Energy
VOCABULARY
energy- the ability to do work or produce heat
Potential energy- energy due to position or composition
Kinetic energy- energy due to the motion of the object and depends on the mass of the object (m) and its velocity
Law of conservation of energy- states the energy can be converted from one form to another but can be neither created nor destroyed
work- force acting over a distance
state function- a property of the system that changes independently of its pathway
temperature- measure of the random motion of the components of a substance
heat- flow of energy due to a temperature difference
system- part of the universe on which we wish to focus attention
surroundings- include everything else in the universe
exothermic- energy flows out of the system
endothermic- heat or energy moves into the system
thermodynamics- study of energy
first law of thermodynamic- the law of conservation "the energy of the universe is constant"
internal energy- the sum of the kinetic and potential energies of all "particles" in the system
calorie- the amount of energy (heat) required to raise the temperature of one gram of water by one Celsius degree
joule- 1 calorie= 4.184 joules
specific heat capacity- the amount of energy required to change the temperature of one gram of a substance by one Celsius degree
Hess's Law- in going from a particular set of the reaction to a particular set of products, the change in enthalpy is the same whether the reaction takes place in one step or in a series of steps
fossil fuel- fuels we get from nature and our environment
petroleum- a think, dark liquid composed mostly of compounds called hydrocarbons that contain carbon and hydrogen
natural gas- consists of methane, but it also contains significant amounts of ethane, propane, and butane.
greenhouse effect- The phenomenon whereby the earth's atmosphere traps solar radiation, caused by the presence in the atmosphere of gases such as carbon dioxide, water vapor, and methane that allow incoming sunlight to pass through but absorb heat radiated back from the earth's surface
second law of thermodynamics- the entropy of the universe is always increasing
energy- the ability to do work or produce heat
Potential energy- energy due to position or composition
Kinetic energy- energy due to the motion of the object and depends on the mass of the object (m) and its velocity
Law of conservation of energy- states the energy can be converted from one form to another but can be neither created nor destroyed
work- force acting over a distance
state function- a property of the system that changes independently of its pathway
temperature- measure of the random motion of the components of a substance
heat- flow of energy due to a temperature difference
system- part of the universe on which we wish to focus attention
surroundings- include everything else in the universe
exothermic- energy flows out of the system
endothermic- heat or energy moves into the system
thermodynamics- study of energy
first law of thermodynamic- the law of conservation "the energy of the universe is constant"
internal energy- the sum of the kinetic and potential energies of all "particles" in the system
calorie- the amount of energy (heat) required to raise the temperature of one gram of water by one Celsius degree
joule- 1 calorie= 4.184 joules
specific heat capacity- the amount of energy required to change the temperature of one gram of a substance by one Celsius degree
Hess's Law- in going from a particular set of the reaction to a particular set of products, the change in enthalpy is the same whether the reaction takes place in one step or in a series of steps
fossil fuel- fuels we get from nature and our environment
petroleum- a think, dark liquid composed mostly of compounds called hydrocarbons that contain carbon and hydrogen
natural gas- consists of methane, but it also contains significant amounts of ethane, propane, and butane.
greenhouse effect- The phenomenon whereby the earth's atmosphere traps solar radiation, caused by the presence in the atmosphere of gases such as carbon dioxide, water vapor, and methane that allow incoming sunlight to pass through but absorb heat radiated back from the earth's surface
second law of thermodynamics- the entropy of the universe is always increasing