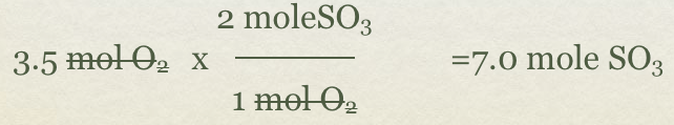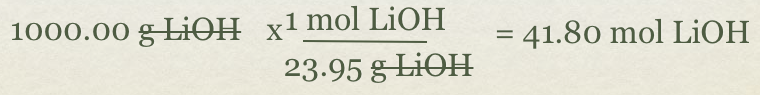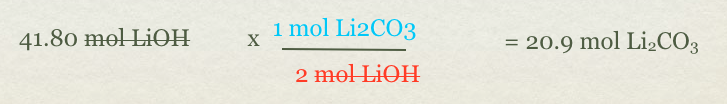Chemical Equations:
Ex.
- Unbalanced SO2(g)+O2(g) ->SO3(g)
- Balanced 2SO2(g)+O2(g) ->2SO3(g)
Stoichiometry
- The process of using chemical equation to calculate the relative masses of reactant and products
Mole to Mole relationship
- All mole to mole relationship comes from balanced equations
- Mole to Mole Conversion is the heart of stoichiometry
Balanced 2SO2(g)+O2(g) ->2SO3(g)
- If we have 3.5 moles of O2(g), how many moles of SO3(g) can be formed?
- Write the given and used the balanced equation to find the mole to mole ratio
Ex.
Steps for Stoichiometry:
1- Identify the given and target compound
2-Balance the equation for the reaction
3- Set up the problem( convert to moles if necessary)
4-Use the mole ratio(s) to calculate the number of moles of the desired compound
5- convert to grams of the desired compound if necessary
Gram to Mole
- LiOH(s) + CO2(g)->Li2CO3(s) +H2O(l)
- Problem how many moles of CO2 will be formed with 1000g of LiOH?
- Step 2 balance equation
- 2 LiOH(s) + CO2(g)->Li2CO3(s) +H2O(l)
- This is important because these coefficient will be needed for the mole to mole conversion.
- 2LiOH(s) + CO2(g)->Li2CO3(s) +H2O(l)
- Problem how many moles of CO2 will be formed with 1000g of LiOH?
- Step 3 set up the problem( convert gram to mol of the same compound)
- We have grams of LiOH but need to go to mol of Li2CO3(s)
- In order to go from one compound to another compound you need moles.
- First, convert grams of LiOH to mol of LiOH using molar mass of the compound
- - 2LiOH(s) + CO2(g)->Li2CO3(s) +H2O(l)
- - Problem how many moles of CO2 will be formed with 1000g of LiOH?
- - Step 4 Convert Mol of a compound to mole of another compound
- - We have mole of LiOH but need to go to mol of Li2CO3(s)
- - In order to go from one compound to another compound you need moles based on the balanced equation.
- - 2LiOH(s) + CO2(g)->Li2CO3(s) +H2O(l)
- - Problem how many moles of CO2 will be formed with 1000g of LiOH?
- - Step 4 Convert Mol of a compound to mole of another compound
- - We have mole of LiOH but need to go to mol of Li2CO3(s)
- - In order to go from one compound to another compound you need moles based on the balanced equation.