Chapter 4 Major Points:
Element symbols: A set of abbreviations. These symbols usually consist of the first letter or the first two letters of the element names.
Compound: a distinct substance that is composed of the atoms of two or more elements and always contains exactly the same relative masses of those elements.
Electron: negative particles in atoms.
Proton: its charge is positive.
Neutron: has no charge.
Isotopes: atoms with the same number of protons but different numbers of neutrons.
Atomic number: the number of protons in a nucleus of an atom.
Mass number: the sum of the number of neutrons and the number of protons in a given nucleus.
Periodic table: this chart shows all of the known elements and gives a good deal of information about each.
Diatomic molecules: molecules made up of two atoms
Ion: taking a neutral atom and adding or removing one or more electrons.
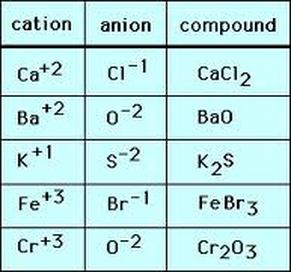
Cation: when one or more electron is lost from a neutral atom.
Anion: when electrons are gained by a neutral atom, an ion with a negative charge is formed.
Ionic compounds: whenever a compound forms between a metal and nonmetal.
Top 5 elements on Earth:
|
Top 5 elements of the body:
|